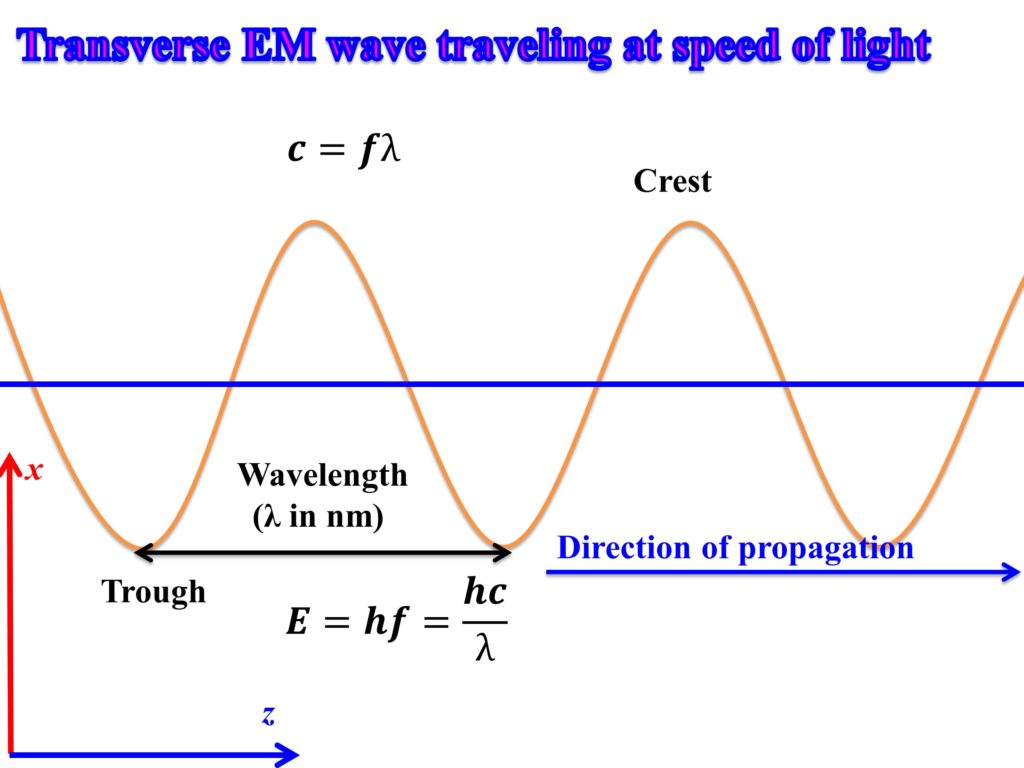
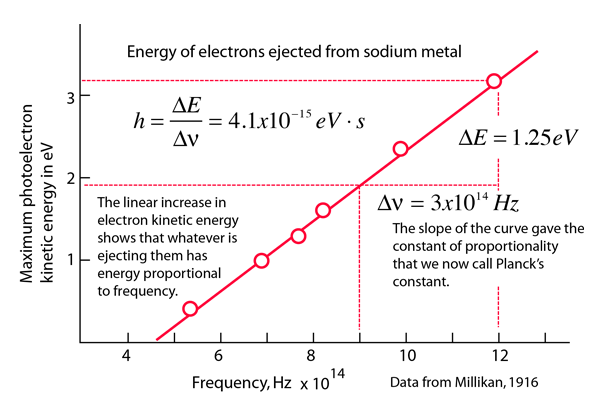
Calculating particle properties of a wave Ch. 12 A light wave consists of particles (photons): The energy E of the particle is calculated from the frequency. - ppt download
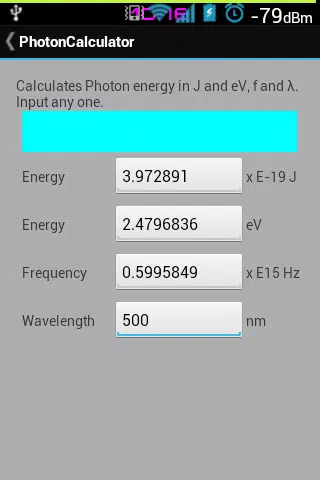
Formula solver for frequency, wavelength, and energy (Photon energy) · Issue #670 · microsoft/calculator · GitHub

OpenStax College Physics Solution, Chapter 29, Problem 10 (Problems & Exercises) | OpenStax College Physics Answers

The energy level diagram of an element is given below. Identify , by doing necessary calculation , which transition corresponds to the emission of a spectral line of wavelength 102.7 nm

Calculating particle properties of a wave Ch. 12 A light wave consists of particles (photons): The energy E of the particle is calculated from the frequency. - ppt download

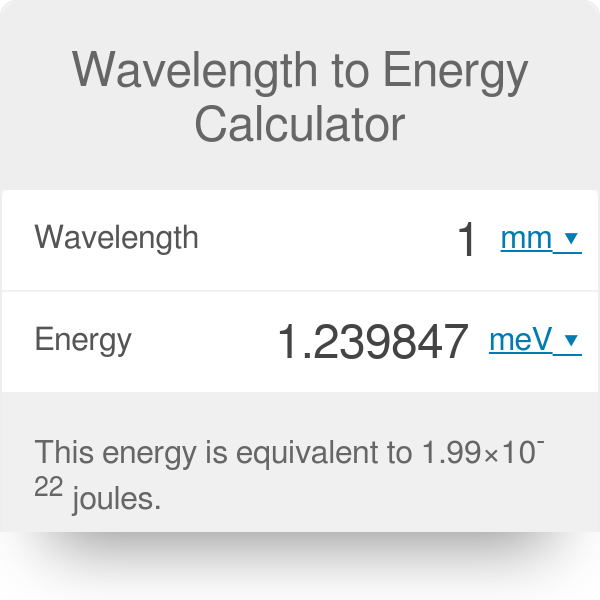
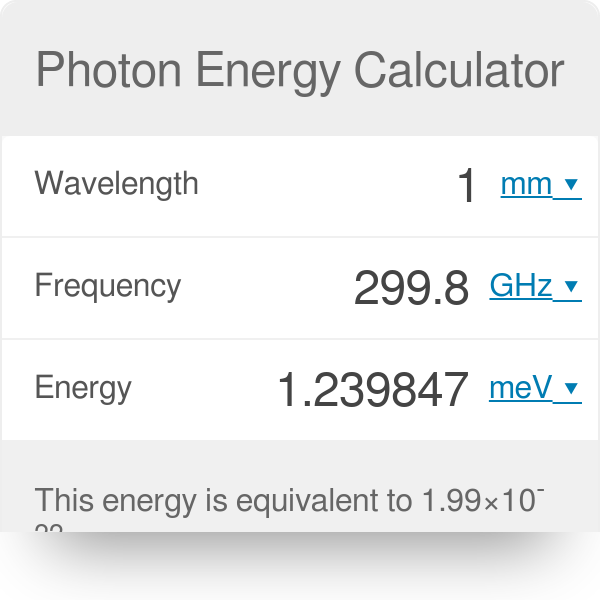
SOLVED:(I) What is the energy range (in joules and eV) of photons in the visible spectrum, of wavelength 400 nm to 750 nm?

Units of E are typically eV and units of λ are typically nm Units of E are typically eV and units of λ are typically nm. Using the hc = ppt download

SOLVED:(I) What is the energy range (in joules and eV) of photons in the visible spectrum, of wavelength 400 nm to 750 nm?